Pre-Filled Saline Syringe Flush Filling Production Line
As a professional manufacturer of prefilled syringe filling lines, Marya produces sterile-grade filling systems for pre-filled saline syringes and disposable catheter flushers. Fully automatic design and closed-loop aseptic control. Our solution helps pharmaceutical companies achieve stable, high-quality sterile production.
- Product Description
- Technical Features
- Technical Parameters
- Optional Systems
- Application Scenarios
- Video
-
1. Introduction
The Pre-Filled Saline Syringe Flush Filling Production Line is an automated production system specifically designed for manufacturing sterile medical consumables such as prefilled plastic syringes and disposable catheter flushers. We have established a fully closed-loop aseptic control system throughout the entire manufacturing process, balancing regulatory compliance, operational stability, and production efficiency. We provide pharmaceutical companies with standardized, verifiable aseptic manufacturing solutions, helping them produce high-quality sterile medical consumables that meet industry standards.
-
1. Compliance & Traceability: Meets GMP Validation Requirements
- The machine is equipped with a PLC-based intelligent control system, enabling real-time monitoring and full traceability of all production parameters, including cleanliness, flushing pressure, and filling volume. It can automatically generate production reports, assisting enterprises in completing the full validation process of IQ (Installation Qualification), OQ (Operational Qualification), and PQ (Performance Qualification). This helps ensure successful compliance with pharmaceutical industry regulations and mitigates production compliance risks.
- The machine features a modular design with a compact structure, facilitating CIP (Clean-in-Place) and SIP (Sterilize-in-Place). It supports the production of various pharmaceutical liquids such as vaccines, biological products, and sterile flush solutions. The machine allows quick changeover between different models of syringes/catheters, meeting the requirements of pharmaceutical enterprises for large-scale, standardized aseptic production. We uphold the essential standards of aseptic compliance, improve production efficiency, reduce operational and maintenance costs, assist manufacturers in producing high-quality sterile medical consumables that meet industry standards, and strengthen their core market competitiveness.
2. Full-Process Aseptic Closed-Loop: Eliminates Secondary Contamination
- Aseptic Filling: The system adopts an isolated closed filling design, ensuring no exposure during liquid transfer. Equipped with a rotary ceramic pump and servo control system, the filling accuracy is controlled within ±1%. It is compatible with prefilled syringes of various volumes ranging from 0.1 to 20 mL, minimizing liquid waste and dosage deviation.
- Integrated Automatic Operation: The machine integrates filling, stoppering, and sealing into a fully automated process with no manual intervention. An online seal integrity detection system ensures leak detection accuracy ≤ 0.01 mL, and non-conforming products are automatically rejected, ensuring that every finished product meets aseptic standards.
- Flexible Adaptability: The modular design offers a compact structure, easy cleaning, maintenance, and upgrade. It supports rapid changeover between different sizes of catheters and multiple types of liquid (e.g., vaccines, insulin, sodium chloride flush solution). Parameters can be customized according to enterprise requirements, accommodating both large-scale batch production and small-batch custom orders.
3. Intelligent Control
- Equipped with an intuitive human-machine interface (HMI), the system enables real-time monitoring of equipment operating status, aseptic environmental parameters, and filling accuracy. Parameters can be stored and retrieved, ensuring full production traceability, facilitating quality control and compliance audits, and reducing operational, maintenance, and training costs.
4. Material Compliance: Meets Medical-Grade Biocompatibility Standards
- All components that come into contact with the liquid are made of medical-grade stainless steel 316L (in compliance with GB/T 1220-2019), with a surface roughness Ra ≤ 0.4 μm. The surfaces are electropolished to provide resistance to acids, alkalis, and chemical corrosion, with no leaching of harmful substances. The materials are validated to ISO 10993 for biocompatibility of medical devices, eliminating the risk of liquid contamination originating from the material itself, thereby meeting the strict material compliance requirements for the production of sterile medical consumables.
-
- Filling range: 0.1ml-20ml;
- Filling mode: Customized (metal pump, peristaltic pump, ceramic pump);
- Filling accuracy: ±1%;
- Silicone oil spraying accuracy: 0.5mg-2mg;
- Capacity: 2000-30000 pcs/hour;
- Pass rate: ≥99.5%;
- Capping pass rate: ≥99.5%;
- Equipment noise: ≤70db;
- Power supply and power rating: 380V 50HZ;
-
- Configurable filling system CIP/SIP system;
- A nitrogen filling unit can be configured to isolate the medicinal solution from oxygen;
- Can be equipped with a vacuum filling system;
- Configurable with Open-RABS isolation protection system and Class 100 air purification laminar flow system;
- The high-performance no bottle no filling, no bottle no capping and linked bottle squeeze stop function can be configured;
- Fully automate the process of filling, stoppering and weighing of pre-filled syringes;
- The automatic calibration of the filling volume can be configured to calibrate the filling volume fully automated;
- Can be equipped with fully closed aseptic isolation (Isolator) systems and bag-in-bag-out (BIBO) systems if needed;
-
This filling line is widely used in core fields such as pharmaceutical manufacturing and medical consumable production. It is particularly suitable for manufacturers of prefilled plastic syringes and disposable catheter flushers. The line can be employed for filling products that demand extremely high aseptic conditions, including clinical catheter flush solutions, vaccines, biological products, and aesthetic skin booster injections.
Years rich Industry Experience
Production Bases
Successful Projects in 60 Countries
Loyal Clientele

Invited to the Embassy of the United Republic of Tanzania in Guangzhou to discuss projects

Tanzanian Vice President Visits Marya Pharmaceutical EPC Project

NBA (Zimbabwe National Biotechnology Authority) conducts FAT at MARYA’s Factory

NOVO NORDISK client Visit

Clients visit the factory's liquid preparation system

Argentine client visits for FAT in MARYA

INTERESTED IN MARYA?
Get In Touch With Us.
We Will Be Happy To Discuss Our Solutions And Services With You.
Contact Now
Full-process Consulting Services
Whether you are preparing to build a new pharmaceutical factory, build a cleanroom that complies with local GMP regulations, or purchase pharmaceutical equipment to develop a new production line, whether it is a separate cleanroom or pharmaceutical machine project, or a one-stop solution, Marya's professional teams in the fields of pharmaceutical technical process, process equipment, decoration engineering, HVAC engineering, pipeline engineering, utility equipment, intelligent automatic control systems, project cost and other fields can provide you with full-process technical consulting services, and solve your needs one-on-one.

2D/3D Design Modeling Services
Marya project design team is composed of professional CAD and BIM engineers with more than 20 years of comprehensive industry experience and can provide you with comprehensive design services, including but not limited to process layout drawings, technical process charts, 3D models and animations, air conditioning system scheme design, construction drawing design, plant/pharmaceutical machine layout drawings, etc.
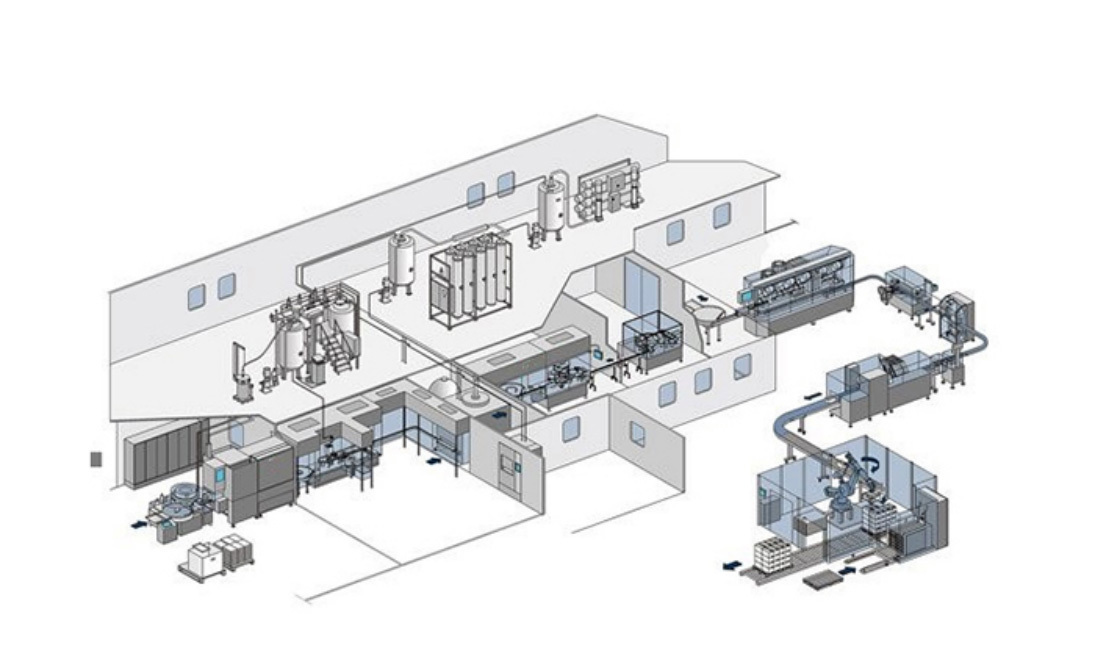
Machine Customization Services
Based on the individual needs of clients in different countries, Marya helps you clarify specific needs, match appropriate models, and customize one stop filling solutions for sterile small-volume preparations, large-volume injections, oral liquids/syrups, solid preparations, etc.

Validation Services (FAT/SAT)
Marya will appoint an engineer with rich project management experience as the person in charge, who will be fully responsible for project management and liaison work. After the equipment is manufactured, our engineers and the buyer's representatives will participate in the FAT test. After the equipment arrives at the buyer's site, it will be installed and debugged. After the equipment reaches optimal operation, the buyer will conduct SAT testing and inspection.
Key words:
Previous:
Contact MARYA Engineers to Claim Your Free GMP compliant Aseptic Pharmaceutical Production Line Solution
Looking for a worry-free, labor-saving, and cost-saving solution? Want to get product catalogs and price lists? Please fill out the form below or send an email and our professional team will contact you within 12 hours.
CONTACT INFO
NO211, North Fute Road, Pudong area, Shanghai, China, 200131




























