Application of Single-Use Technology in Aseptic Filling Process
- Categories:Knowledge Center
- Author:Marya
- Origin:original
- Time of issue:2021-04-13 17:09
- Views:
(Summary description)Application of Single-Use Technology in Aseptic Filling Process, including four aspects of terminal sterilizing filtration, aseptic filling background environment, aseptic transmission system, aseptic filling bag.
Application of Single-Use Technology in Aseptic Filling Process
(Summary description)Application of Single-Use Technology in Aseptic Filling Process, including four aspects of terminal sterilizing filtration, aseptic filling background environment, aseptic transmission system, aseptic filling bag.
- Categories:Knowledge Center
- Author:Marya
- Origin:original
- Time of issue:2021-04-13 17:09
- Views:
After more than 30 years of development, Single-Use Technology(SUT in short) has covered all operating units of the whole bio-pharmaceutical process, from upstream cell culture to downstream purification till final filling.
Compared with traditional stainless steel system used repeatedly, SUT has the advantages of flexible operation, high safety, shortened production cycle and reducing initial input cost, to meet the requirement of regulatory departments for pharmaceutical enterprises with maximum limit to reduce the risk of cross-contamination and reduce cleaning verification requirements.
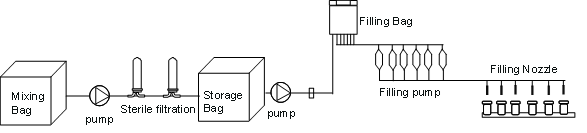
Figure 1: process flow chart of disposable filling system
The preparation filling process includes semi-finished product preparation, terminal sterilization and filtration, aseptic transmission and aseptic filling distribution. Disposable filling system is a pre-verified, pre-installed and pre-sterilized aseptic transmission unit, including disposable liquid distribution bag, liquid storage bag, high buffer bag, disposable bag filter, filling pipeline, disposable aseptic connector, breaker, disposable filling needle and other components.
1.Terminal Sterilizing Filtration ————-———————————————————————————————-————————
According to the appendix to GMP, the guidelines for sterilization and filtration technology and application stipulate that the release of bacterial gas or liquid should be taken into account when designing filter location, and confirm the area and location of filter installation based on the batch of products, the length of pipeline, the convenience of installation and sterilization, etc.
After filter is used, the integrity of the filter must be tested and recorded immediately with appropriate methods. Before use of sterilization filter, a risk assessment should be conducted to determine whether an integrity test should be carried out and whether it should be carried out before or after sterilization.
If a disposable filtration system is used and pre-use integrity testing or flushing is required, the following factors should be taken into account in the design: pressure resistance of upstream connection pipeline, sterility of downstream, and downstream can provide sufficient space (such as installing sterile barrier filters or corresponding volume aseptic bags) for exhaust and drainage.
2.Aseptic Filling Background Environment ————-———————————————————————————————-————
The aseptic filling production of biological agents must be carried out in the environment of Class100 (ISO5,Class A). According to different background environment, the local environment of filling machine can be divided into LAF (Laminar Airflow), RABS(Restricted Access Barrier System, Open or closes) and isolator.
ORABS is an open restricted access barrier system, which is the most commonly used filling barrier at present. It is equipped with a glass door metal frame and intervention gloves, and a static pressure box to provide one-way vertical air flow at top, and supply air at top is discharged from the bottom and returned through B-class background.

Figure 2: ORABS
With the increasing maturity of Isolator technology and increased risk awareness of production staff, the isolator has been widely used in a C-class or D-class clean area. All personnel operation and material / tool transfer during production can not destroy tightness of the system. The isolator should be automatically cleaned with VHP before use, and its repeatability and controllability need to be verified.

Figure3: Isolator
3.Aseptic Transmission System ————-———————————————————————————————-—————————
The system runs through the whole aseptic filling process, and through-wall cross-level transport system is a common tool. Many steps such as cleaning and sterilization are involved in a traditional way, but one-time cross-level wall-crossing system can solve this problem more safely and conveniently. The system provides a simple, safe and reliable scheme for transfer and transportation of a large number of fluids in different function rooms. The whole system realizes aseptic transmission of multi-pipeline fluid through a stainless steel channel and pre-sterilized disposable components, and meets a variety of process requirements: 1, the same level through wall 2, cross-level through wall 3, high activity and low activity transport (ADC, etc.) 4, toxic and non-toxic transmission (virus vaccine, etc.).
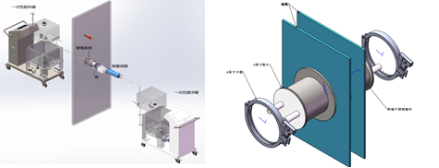
Figure 4: Aseptic Transmission System
How to quickly transfer items (liquid, aluminum-plastic covers, rubber plugs and tools, etc.) to or from the controlled environment in isolator, RTP(Rapid Transfer Port) is an ideal choice. RTP system consists of two parts: α port and β port. α port is installed on isolator door and is normally closed, while β port components include a flange, a seal and a door, which can be divided into three types: stainless steel transfer barrel, plastic transfer barrel and disposable transfer bag. After β port components is connected with α port, the operator opens RTP door from inside of isolator by gloves, and then transfers pre-sterilized material to inside of isolator, and the whole process ensures isolation from outside.

Figure 5: RTP (Rapid Transfer Port)
4.Aseptic Filling Bag ————-———————————————————————————————-——————————————
The disposable aseptic filling bag includes high buffer bag, filling pipeline and filling needle. The high buffer bag is a liquid temporary storage bag in filling process, which feedback start and stop of peristaltic pump through weighing sensor on weighing, so as to continuously replenish liquid, maintain steady liquid level height and inlet pressure, and finally ensure good filling accuracy.
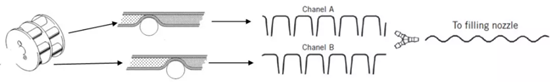
Figure 6: Diagram of Filling Pipeline
The filling line includes liquid inlet pipe in front of peristaltic pump , pump pipe and outlet pipe at back of the pump, in which the pump pipe especially refers to a part of hose extruded in filling pump (that is, peristaltic pump), which is an important factor to determine filling accuracy. Each filling pump usually has two rollers, each roller is composed of 4-8 rolling pulleys, each pump tube passes through a roller to form a channel, at last two hoses converge into a channel. To counteract pulse through superposition of peaks and troughs of two channels. The liquid discharge mode of filling bag is also a factor affecting unstable filling accuracy, and the commonly used ship type and octopus type can meet the requirement of uniform liquid separation. In addition, inverted trapezoidal bag design can minimize waste of liquid medicine.


If you want to know more about our aseptic filling process, please click this link https://www.marya.com.cn/product/Aseptic-pre-filled-syringe-and-seal-line-107.html
CONTACT
Shanghai Office: NO211, North Fute Road, Pudong area, Shanghai, China, 200131
Changsha Office: Room 23069, Block 6, Forte Star Shine World, Yuhua District, Changsha, Hunan province
Factory 1: Hunan Marya Technology Co., Ltd. Address: No. 7 Plant, SANY Industrial Park, No. 1, SANY Road, Changsha Pilot Free Trade Zone, Hunan Province, China, 410100
Factory 2: No 555, Yupan Road, Weitang Town, Xiangcheng District, Suzhou, Jiangsu Province, China
Factory 3: 15-A, Venture Capital Industrial Square, Yanghe Road, Suzhou Industrial Park
Tel:+86-13507449007
E-Mail:info@marya.com.cn
SEARCH
Copyright © 2020 Shanghai Marya Pharmaceutical Engineering & Project Co., Ltd 沪ICP备2021002344号-1
- +86-13507449007
- 返回顶部





